1. Introduction
The currant and gooseberry genus RibesL. (Grossulariaceae) includes about 200 species of deciduous, evergreen, shrubs that grow in the colder and temperate parts of North America, Europe, Asia, and South America [1-3]. In Flora of Turkey, the genus Ribes is represented by eight species. Seven Ribes species native to Turkey,these are as follows R. biebersteinii Berl. ex. DC.,R. nigrum L., R. uva-crispa L., R. alpinum L., R.orientale Desf., R. multiflorum Kit. ex Romer & Schultes, R. anatolica Behçet (endemic) and commonly cultivated species is R. rubrum L. [4-6]. R. nigrum species has been evaluated VU (Vulnerable) category by IUCN (International Union for Conservation of Nature) Red List Categories [7]. Ribes species usually grow in the northeastern Anatolia in Turkey and all are popularly known as “frenk üzümü, it üzümü or bektaşi üzümü” [8].
Ribes species contain many active components such as flavonoids [9-11], tannins [12], biphenyls [13], anthocyanins and nitrilecontaining compounds [9], polyunsaturated fatty acids [14] and aromatic compounds (including terpenes, esters and alcohols)[15].
The leaves and fruits of Ribesnigrum are widely used in the treatment of gastrointestinal disorders [16], cough, common cold [17], diabetes [18], and rheumatic arthritis in the worldwide [19]. The leaves and buds of R. nigrum due to their characteristic color and excellent flavor, have also used some applications as a raw material for the food and cosmetic industries [15]. Furthermore, fruits of some Ribes species are used for preparing juice and jam [20]. R. nigrum is an economically important crop in northern Europe and Australia. The leaves of Ribesnigrum are recorded as adjuvan in the treatment of rheumatic conditions in ESCOP Monographs and as increase the amount of urine to achieve flushing of the urinary tract as an adjuvant in minor urinary complaints in European Medicines Agency [21,22]. Leaves of R. nigrum, R. rubrum and R. orientale have been utilized in folk medicine in Turkey due to their diuretic and diaphoretic properties [23]. Furthermore, leaves of R. nigrumare used as an herbal tea in Elazığ province in Turkey [24]. Fresh or dry fruits of R. biebersteinii are used as food and against anaemia in Erzurum (Turkey) [25,26]. Fresh fruits of R. uva-crispa have been utilized as laxative, diuretic, gastric and appetiser in Turkish traditional medicine [23].
The plants and their secondary metabolites have various biological activities such as antimicrobial, antitumor, antioxidant etc. Antioxidant plants or compounds are important as antioxidants for the prevention and treatment of free radical-related disorders [27]. In addition, the natural products derived from medicinal plants have proven to be an abundant source of biologically active compounds, many of which have been the basis for the development of new lead chemicals for pharmaceuticals [28].
Many papers are presented the chemical composition, biological activities and food-stuff properties of the fruits of Ribes nigrum and R. rubrum, especially; however, many studies on leaf and branch of the other Ribes species have not been reported [29-33]. In this study, we investigated of total phenolic content and antioxidant activity of the Ribes species of leaves and branches growing in Turkey. To the best of our knowledge, there is no antioxidant activity study on the other species in Turkey.
2. Materials and Methods
2.1 Chemicals
Methanol (Sigma), Dichlormethane (Sigma), Ethyl acetate (Sigma), n-Buthanol (Sigma), Folin-Ciocalteu reagent (Molychem 31740), Gallic acid (Riedel de Haen), DPPH● reagent (Sigma, D9132), Phosphate buffer (Biomatik A3602), Ferric cloride (Sigma F1513), Ascorbic acid (Aldrich 25,556-4), Thiobarbituric acid (Sigma- Aldrich T5500), Butylated hydroxytoluene(Sigma-Aldrich B1378), Brain extract (B3635), Rutin (R5143), Proply gallate (P5,330-6), Chlorogenic acid (C3878), TLC plates (Merck).
2.2 Plant materials
The leaves and branches of Ribes species used in this study were collected during the flowering or fruity periods from Aegean,Black see, East and Inner Anatolian regions of Turkey. Voucher specimens are deposited at the Ankara University Faculty of Pharmacy Herbarium (AEF). The collection locations of the research material are listed in Table 1.
2.3 General experimental procedures
5 g of leaf and branch of each species were powdered and macerated with 100 mL of MeOH during 3 days for 8 h at 60°C. The extracts were filtered, after which the filtrate is evaporated to dryness in the rotary vacuum evaporator at 40°C .25 g of leaf and branch of each species were extracted with 200 mL of methanol in the same way. This residue was dissolved in 100 mL water, then partitioned three times with dichloromethane (3×100 mL), EtOAc (3×100 mL) and n-BuOH saturated with water (3×100 ml). All the three fractions were evaporated to dryness within the rotary vacuum evaporator. 10 g leaf and branch of each species were powdered and added to 100 mL distilled water, after which boiled for 30 min. The extracts were filtered when hot was and after that lyophilized in a Freeze Dryer (Christ Gamma 2-16 LSC, Germany).
The preliminary phytochemical screening of various active compounds (tannins, flavonoids, coumarins, saponins etc.) from the leaves and branches of Ribes species were performed.
2.4 Determination of total polyphenols
The total polyphenol content of Ribes extracts was determined colorimetrically by Folin-Ciocalteau reagent as gallic acid equivalents [34-36].The assay was performed by mixing 250 μL of Folin-Ciocalteu’s reagent, 50 μL of the samples and 500 μL of 20% water solution of Na2CO3. Mixtures were vortexed and completed with water to 5 ml. Absorbance of the mixture was measured at 765 nm (Shimadzu UV- 1800) after incubation at room temperature for 30 min. A standard curve was created with gallic acid. The total polyphenols were given as gallic acid equivalents (GAE) and expressed in mg GAE/g extract (dw)±standard deviation (SD).
2.5 Antioxidant activity
2.5.1 Antiradical activity
Free radical scavenging activity of Ribes extracts has been investigated by two different DPPH test:
2.5.2 Qualitative DPPH●
DPPH assay with rapid thin layer chromatography (TLC) were used to evaluate the antioxidant activity of the extracts of Ribes species due to free radical scavenging. DPPH●, a purple-colored stable free radical that was reduced in toyellow-colored diphenyl picryl hydrazine. When it is sprayed onto a TLC plate, active compounds appear as a yellow zones on a purple background. 3 μLRibes extracts was applied to the silica gel TLC plates (Merck, Darmstadt, Germany), after which were sprayed with 0.2% DPPH reagent in MeOH, stayed at room temperature for 30 min after spraying [37,38].
2.5.3 Quantitative DPPH●
Free radical scavenging activity of Ribes species extracts was done according to the method of Brand-Williams et al. [39]. 0.1 ml extract (in seven different concentration 10 mg/ml, 5 mg/ml, 2.5 mg/ml, 1.25 mg/ml, 0.625 mg/ml, 0.3125 mg/ml, 0.15625 mg/ml) was added to 2.9 ml DPPH solution (10−4M in methanol) as free radical source. The mixture was shakenand allowed to stand in water bath at 30°C for 30 minutes. Each extract ofthe absorbance was read at 517 nm with a model UV-1800 spectrophotometer (Shimadzu). Propyl gallate was used as positive control. Propyl gallate, rutin and chlorogenic acid was used as a reference compounds. Results were given as radical scavenging activity percentage (%) of the DPPH, was calculated by the formula [(Ao −As)/Ao]×100, where Ao is the absorbance of the control, and As is the absorbance in the presence of the sample or standard. The IC50(half-maximal inhibitory concentration) values of the extracts were determined by linear regression analysis. The assay was performed in triplicate.
2.5.4 Anti-lipid peroxidation activity
The thiobarbituric acid (TBA) assay was used to evaluate the efficacy of the extracts in protecting liposomes from lipid peroxidation [38]. Seven different concentrations (0.016–1 mg/ml) of the extracts was prepared in this test.Propyl gallate, rutin and chlorogenic acid was used as a reference compounds in seven different concentrations (0.000064–1 mg/ml). Rutin and chlorogenic acid were prepared in the same concentration range and studied. Tested of extract was added to 0.2 mL brain extract, 0.5 mL phosphate buffer, 0.1 mL ferric cloride, and 0.1 mL ascorbic acid. The mixture was incubated 37°C for 20 minutes. After incubation, 0.1 mL 2 % BHT, 0.5 mL of 1 % TBA and 0.5 ML of 25 % HCL were added in the mixture. The mixture was shaken and incubated 85°C for 30 minutes. After cooling, 2.5 mL n-butanol was added. The mixture was centrifuged and then absorbance of the extracts was read at 532 nm using UV-1800 spectrophotometer. The assays were performed in four replicates. The IC50 (half-maximal inhibitory concentration) values of the extracts were determined by linear regression analysis.
3. Results
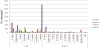
In this study, antioxidant activities of different extracts prepared with different polarities of solvents prepared from the leaf and branch of Ribes species of Turkish origin were evaluated by using in vitro methods. Free radical scavenging activities and effect of liposome lipid peroxidation of the extracts were evaluated. In addition, the total phenolic contents were assesed of the extracts. Preliminary phytochemical analysis showed the presenceof flavonoid and tannin in the leaves and branches of Ribes species. Total phenolic contents of the extracts were evaluated by using Folin-Ciocalteau reagent as colorimetric.The results of total phenolic contents are given Table 2. The highest total phenolic content were determined in ethyl acetate (1067.19 to 345.31mg GAE/g dry weight) and n-buthanol extracts (217.50 to 687.81 mg GAE/g dry weight)of the leaves and branches of the species. Low total phenol content was determined in the dichloromethane extracts of Ribes species. The highest total phenolic content was present ethyl acetate extract of R. uva-crispa leaves (1067.19mg GAE/g dry weight), followed by ethyl acetate extract of R. orientale leaves (951.56 mg GAE/g dry weight). In Qualitative DPPH● assay, both parts of whole Ribes species of theethyl acetate and n-buthanol extracts were observed to have high radical scavenging effect compared with the standards (propyl gallate, chlorogenic acid and rutin). Quantitative DPPH● assay results were present as IC50 values (μg/mL) in Figure 1. Apart from R. multiflorumand R. anatolica extracts, high DPPH radical scavenging activity was determined in the extracts of Ribes species. The standards (propyl gallate, chlorogenic acid and rutin) were exhibited high radical scavenging activity than the extracts. TBA assay results of Ribes species extracts were present as IC50 values (μg/mL) in Figure 2. The highest antioxidant effect of the water and methanol extracts was observed, whereas the dichloromethane extracts no antioxidant effect was observed. The highest activity was shown in the water extracts of leaf and branch (IC50: 0.30 and 0.44 μg/mL, respectively) of R.orientale. Ethyl acetate extracts of R. rubrum branch and leaf were not determined antioxidant effect on liposome. Water extract of R. uva-crispa branch and methanol extract of R. anatolica branch were exhibited considerable antioxidant activity in TBA assay. Most of the plant extract was exhibited antioxidant activity on liposome compared with chlorogenic acid and rutin. A correlation between the values obtained from DPPH● assays and total phenol content was observed. However,this correlation was no shownbetween the values obtained from TBA assay and total phenol content.
4. Discussion
In this study, the antioxidant activities of extracts in the different polarities obtained from eight Ribes species were evaluated. Furthermore, basically phytochemical screening and the total phenolic contents were determined of these eight species.
The genus was screened for the presence of flavonoids, alkaloids, saponins, anthocyanins, tannins, cyanogenic glycosides, anthraquinones, coumarins and volatile oil. The positive test was obtained for flavonoids and tanins of whole Ribes species of the leaves and branches. However, volatile oil was detected in leaves of all species only. These results are agreed with the references [9-12,14].
Total contents of the phenolic compounds in the extracts were determined by the Folin-Ciocalteu’s method as gallic acid equivalents (GAE). According to the results obtained from the determination of total phenolic contents, it was generally found that the water, MeOH, EtOAc and BuOH extracts include more phenolic contents than the DCM extracts.
According to the literature surveys, antioxidant activity studies carried out especially on Ribes species fruits. The most studied species was detected as R. nigrum cultivars. In the reports, the high values of antioxidant activity were observed in the fruits of R. nigrum [40,41]. Tabart et al. [29,30,32] were carried out antioxidant activity studies on different parts of Ribesnigrum cultures (for example bud, leaf, fruit) and obtained effective results from this studies. Ribes fruit grown in organic system was exhibitedsignificantly higher total phenolics and antioxidant activity (DPPH and ferric reducing antioxidant power assays) than fruit grown in conventional system [42]. Fruits of R. nigrum showed higher values for antioxidant capacity than fruits of R. rubrum [43-45]. The results of these both species in our study are consistent with the literature data.The antioxidant activities of R. multiflorumfruits which grow in Turkey was evaluated and observed to have a strong antioxidant effect [46]. In ourstudies, the extracts of prepared from leaves and branches of R. multiflorum were observed effective antioxidant activity. In a study conducted Iran, methanol extract of R. biebersteinii leaves was determined to pose strong radical scavenging activity [31]. Similar results were observed in our studies for this species. R. uva-crispa fruits was not shown significant effect in the antioxidant activity studies conducted on berries [47,48]. The leaves and braches of water and MeOH extracts were found significant activity for R. uva-crispa.
R. alpinum, R. orientale and R. anatolica of antioxidant activities were evaluated for the first time with our studies. Antioxidant activity study was not observed on branches of Ribes species, previously.
5. Conclusion
In this study, antioxidant activity of leaf and branch of Ribes species which grow in Turkey was demonsrated for the first time. R. orientale leaf and branch of water extracts exhibited the highest antioxidant activity in the in vitro assays. Polyphenols such as flavonoids and tannins which are a class of secondary plant metabolites with potent antioxidant and chelating features. The ability of polyphenols to inhibit free radicalmediated events is administered by its chemical structure. Many dietary polyphenolic compounds derived from plants are more powerful antioxidants in vitro than vitamin C or E [49,50]. These results revealed that R. Orientale may be a good antioxidant source of natural origin. Further studies are required to identify the bioactive compounds responsible for the activity in Ribes species.
Competing Interests
The authors have no competing interests with the work presented in this manuscript.
Author Contributions
All the authors substantially contributed to the study conception and design as well as the acquisition and interpretation of the data and drafting the manuscript.