1. Introduction
Although caesarean section (CS) is an effective procedure in preventing maternal and perinatal mortality and morbidity, it is not without risks and it has been associated with short- and long-term complications [1]. Many studies show that women with CS history have a higher risk of the following complications: haemorrhagic disorders, abnormal placentation, uterine rupture, stillbirth, and repeated caesarean sections [2].
Additionally, the CS scar still remains the main risk factor for a morbidly adherent placenta [3] and several studies have concluded that multiple repeated CS deliveries imply an increased risk of abnormal placentation with maternal and fetal morbidity, exponentially increasing with the number of prior caesareans performed [4]. Morbidly adherent placenta was first described in the 20th century and reported in the 1930s [5]. Initially thought to be a rare pregnancy complication, a substantial increase of cases of placenta accreta occurred in the last 20 years, amounting to 3/1000 deliveries [6]. Regarding the placenta accreta occurrence, we found a dose-dependent relationship with prior CSs ranging from a seven-fold increased risk after one prior CS to a 56-fold increased risk after three or more CSs. If considered the association between placenta previa and accreta, a prospective study by Chattopadhyay et al. observed that placenta praevia complicated 2.54% of cases with one previous caesarean section, compared with 0.44% of cases with no scar, that is a 5-fold increase and, in particular, after one caesarean section, placenta praevia was complicated by accretion in 10% of cases and after two or more this was 59.2% [7]. In a metaanalysis of 1996, assessing the risk of placenta previa with prior caesarean delivery, the risk of placenta previa ranged from 0.28 to 2.0% [8]. Creanga et al. observed that caesarean deliveries morbidity declined modestly from 2000-2011, but placenta accreta became an increasingly important contributor to repeated cesarean delivery morbidity [9]. Clark et al showed that the risk was significantly increased with the fifth and sixth cesarean delivery compared with risk after the third procedure [10,11].
Abnormal placentation has been classified into accreta, increta and percreta, basing on the myometrial invasion depth: superficial, deep, and through the uterine serosa, respectively. The bigger the invasion, the greater the risk for haemorrhage and maternal morbidity [12]. Its etiopathogenesis is usually a consequence of an intensive production of growth and vascular factors by trophoblastic tissue, leading to the formation of a rich vascular anastomotic system among the urinary bladder, uterus and vagina and involving the superior, medial, inferior vaginal and lower vescical arteries [13]. Placenta percreta is one of the most serious variants of placentation and is associated with a significantly higher maternal morbidity than the other clinical conditions [14].
Placental attachment disorders (PAD) clearly show the following documented risk factors: previous uterine surgical procedures, placenta previa, previous caesarean sections and maternal age [12]. Placenta accreta is respectively found in 0.24%, 0.31%, 0.57%, 2.13%, 2.33%, and 6.74% of women undergoing their first, second, third, fourth, fifth and sixth or more caesarean deliveries [4]. According to an etiopathological theory, there is a resulting un-stretched uterus and a mechanical damage to endometrium and myometrium that will not allow a correct placental migration [15]. Recent evidences focus on caesarean scar pregnancy (CSP) as the precursor of PAD [16].
During the prenatal period, to distinguish between these different clinical entities, is not particularly easy. Ultrasound (US) and Magnetic Resonance Imaging (MRI) are used to obtain a prenatal diagnosis. Standard transabdominal US is a reliable tool for diagnosing invasive placentation and is the primary one for the antenatal diagnosis of a morbidly adherent placenta. The gestational sac located in the lower urine segment in the first trimester of pregnancy has been correlated with an increased incidence of placenta accreta in the third trimester [17]. Several other US features showed an association with a higher risk of PAD such as: placental lacunae (irregular vascular spaces resulting in a Swiss cheese appearance), retroplacental myometrial thickness less than 1 mm, loss of the normal hypoechoic retroplacental zone and anomalies of the bladder-myometrium interface. US sensitivity was 100%, with the presence of placental lacunae found during the second trimester, having the highest sensitivity (93%) [18,19]. Diagnostic accuracy appears to improve using a combination of US techniques (grey scale and color Doppler), instead of using only one ultrasound finding. Employing the three grey-scale signs (clear space, bladder line interruption, lacunae) and 2D-3D color Doppler signs (tortuous confluent vessels crossing placenta width and hypervascularity of the uterine serosa bladder wall) Cali et al. found out that all 41 women with PAD had two or more such signs [20]. MRI is usually the imaging technique and has been reported to provide an accurate anatomical description of the area invaded by the placenta, particularly when it has a posterior lying, or in the cases when its ultrasound findings are difficult to interpret [21]. In women with anamnestic risk, US signs of MAP (Morbidly Adherent Placenta) should be searched for and serial follow-up scans should be performed from the 28th week of gestation. These measures are needed in order to accurately predict the extension of the invaded area and plan the best surgical treatment [22]. Although prenatal imaging may help in detecting PAD, it is not entirely certain whether it aids in determining the optimal surgical approach or whether it can predict intraoperative complications. The optimal surgical approach to PAD has not yet been established and depends upon several factors such as: an experienced team, specific surgical skills and hospital resources. No randomized controlled trial comparing different surgical strategies in cases of PAD has been published yet [22].
The optimal management of this condition requires a multidisciplinary team approach headed by the obstetrician, involving the anaesthetist, the diagnostic and interventional radiologist, the haematologist, the urologist, the vascular surgeon and the neonatologist. Indexing to tertiary referral centres, where facilities are available for radiological interventions, blood products and cell savers should be considered in all suspected cases of placenta previa accreta, increta and percreta, especially in women who refuse blood transfusions [23]. In the cases of prenatal diagnosis of placenta accreta, and expecially increta and percreta, caesarean hysterectomy should be the planned treatment (avoiding placental removal repeated attempts). Anyway, there are now multiple studies and case reports for the conservative management of abnormal placentation. This management may have a limited role in carefully selected patients who desire future fertility. In some cases, the placenta is left in situ for resorption or uterine artery embolization followed by expectant management and adjuvant medical therapy is performed, including Methotrexate, Misoprostol, Mifepristone and GnRH analogues [24,25]. However, the complications associated with leaving the placenta in situ have haemorrhagic and infectious nature, associated with a very high risk of subsequent hysterectomy until nine months after delivery [26], while the most common affected pelvic structure is the urinary tract [27].
2. Case Presentation
A 27-year-old woman (gravida 9, para 6), with a body mass index of 24.7kg/m2, was immediately admitted to our Emergency Department for pelvic pain due to the high risk of preterm delivery at 31 weeks of gestational age. The patient had previously performed eight uterine surgical procedures from 2004 to 2016, consisting of six previous caesarean sections, one of them complicated by preterm abruptio placentae, and two uterine dilations and curettage for two spontaneous abortions.
She has performed only two ultrasound examinations in the second trimester of pregnancy, where a condition of an abnormally low-lying placentation was detected, but no concerns about an abnormally attachment were made and no additional diagnostic examinations were performed.
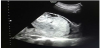
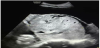
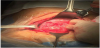
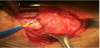
Her past medical history was not significant. Taking into consideration the gestational age, the woman was strictly monitored and underwent the prophylactic double-dose of corticosteroids (betamethasone 12mg per im on the day 1 and 2). She was submitted to an ultrasound examination (Figure 1 and Figure 2) which confirmed the central placenta previa diagnosis. Furthermore, there was an abnormal placental attachment directly on the uterine serosa layer, without myometrium and in direct proximity to the urinary bladder serosa with no clear appearance of a cleavage plan and focal areas of placenta percreta. No blood discharge was present and the uterine cervix was shortened and softened by funnelling, so we decided to urgently perform a caesarean section (Figure 3 and Figure 4) with subtotal hysterectomy. As soon as the Pfannenstiel incision was performed, it was clear that the bladder peritoneum was seriously adherent to the membranes of the anterior uterine wall which appeared incomplete and dehiscent with the placenta located on the uterine sierosa layer. After a trans-placental extraction of a female baby in transverse situation, it was not possible to detach the placenta from the residual uterine wall. The subtotal hysterectomy was performed. The major difficulty was the separation between the uterus and the bladder serosa due to the adherence of the surgical plane. The newborn was in good general conditions, weighed 1,610 gr with an Apgar score of 8/9 after the 1st and 5th minute, respectively. A single-unit blood transfusion was administered to the mother during the surgical intervention for a chronic anaemic condition (preoperative haemoglobin level of 9.2 mg/dL). No postoperative complications occurred and the patient was discharged from the hospital on the 6th after delivery. The pathologist confirmed the bladder mini-invasion by the placenta percreta, located in several areas.
3. Discussion
Risk factors for peripartum hysterectomy in women with a diagnosis of placenta previa are associated with previous caesarean section(s), a complete placenta previa, a placenta accreta, increta or percreta, an anterior placenta, antenatal anemia and a delivery before the 34th gestational weeks of pregnancy, associated to all the myometrial damages caused by surgery, involving the uterine cavity and damaging of the endometrium-myometrial interface (manual removal of the placenta, uterine curettage, endometritis, uterine scars) [28]. Prior post-partum haemorrhage is another risk factor related to an increased six-fold prevalence of abnormal placentation [29].
Central placenta previa and PAD are potentially life-threatening obstetrical conditions, who require frequent monitoring and risk evaluation during the whole gestational period from the first trimester. Additionally, a multidisciplinary approach is required during delivery in order to minimize haemorrhagic maternal complications, peripartum hysterectomy and significantly reduce neonatal morbidity and mortality. There are no highly specific ultrasound signs for a reliable diagnosis of PAD, but the findings of placental lacunae, placental bulge and a focal placental exophytic mass were recurrently connected with a deeper myometrial invasion [30]. On top of that, grey scale ultrasonography has shown a sensitivity and specificity of 88 and 90% respectively for the diagnosis of placenta previa accreta or with a deeper invasion, when performed by skilled ultrasound operators [31]. Inversely, when the diagnosis occurs incidentally during the emergency condition such as premature delivery, there is an increased risk of maternal haemorrhagic complications. The recommended action in these cases is a caesarean hysterectomy. However, the gold standard should be a planned caesarean section with the organisation of a multidisciplinary team, in order to better individualise the patients’ treatment, to further evaluate the necessity of a hysterectomy and to adequately manage haemorrhagic complications [28,31].
4. Conclusion
We conclude that the gold standard approach for the treatment of PAD and MAP is first of all an early diagnosis, since the second and, where possible, the first trimester of pregnancy. Morbidly adherent placenta should be ever prenatally identified (or suspected) in order to medically and/or surgically manage it. Treatment options should be tailored basing first of all on patient preferences when possible and when expressed, on Obstetricians experience and last but not the least, on the availability of a multidisciplinary team in order to put maternal and fetal safety first.
Competing Interests
The authors declare that they have no competing interests.