1. Introduction
Prediction of cardiac event risk is important for determination of an optimal treatment strategy in patients with coronary artery disease (CAD). In 1998, Wilson et al. [1] reported the first risk score, i.e. a Framingham risk score, developed on the basis of results of a largescale epidemiological study to predict future cardiac events in the United States. The Framingham risk score provides the risk of CAD over 10 years in a Caucasian population according to categorical data on sex, age, low-density lipoprotein (LDL) cholesterol, high-density lipoprotein (HDL) cholesterol, blood pressure, diabetes mellitus (DM) and smoking. This algorithm is a highly recommended tool to predict the risk of cardiac events in the Guideline on the Assessment of Cardiovascular Risk published by American College of Cardiology and American Heart Association in 2013 [2]. However, this algorithm is applicable to limited ethnic groups but not to especially Asian races, and also is based on classical risk factors derived from epidemiological research.
Recently, the remarkable development and technical innovation of coronary imaging has provided various types of diagnostic imaging, which are widely used against CAD patients. Especially myocardial perfusion single photon emission computed tomography (SPECT) is a modality with abundant evidence for management of CAD patients and cardiac event risk stratification. In this article, the usefulness of nuclear cardiology for prediction of cardiac event risk and risk stratification in Japanese patients with CAD is summarized on the basis of Japanese evidence and results of our clinical research.
2. Cardiac event risk stratification using myocardial perfusion SPECT
There are many reports on the usefulness of myocardial perfusion SPECT for evaluation of ischemia and prediction of cardiac events [3-5]. Hachamovitch et al. [6] demonstrated a correlation between cardiac event risks and defect scores obtained from stress myocardial perfusion SPECT in a prognostic follow-up study in approximately 5,000 patients on the basis of their plentiful database. They calculated summed stress scores (SSS) for the defect scores and classified them into four categories, i.e. normal (SSS < 4) and mildly (SSS 4 to 8), moderately (SSS 9 to 13), and severely (SSS > 13) abnormal, to estimate the incidence of cardiac events in each category. As a result, the incidence was lower (0.7%/year) in the normal group and significantly increased depending on the severity of SSS. Since the report was valued as evidence for prognostic prediction in the United States, the cardiac event risk stratification with myocardial perfusion SPECT is highly placed in the ACCF/AHA/ACP/AATS/PCNA/SCAI/ STS Guideline for the diagnosis and management of patients with stable ischemic heart disease [7].
In Japan, a large-scale multicenter prospective J-ACCESS (Japanese Assessment of Cardiac Events and Survival Study) [8] has been conducted since 2001 to evaluate the prognostic value of myocardial perfusion SPECT in patients with suspected or confirmed CAD. The results demonstrated that patients with normal SSS have a good prognosis, an increase in SSS is associated with an increase in the risk of cardiac events, and the risk of cardiac events is stratified with the severity of SSS also in Japanese patients. Furthermore, analyzing the results obtained from the J-ACCESS sub-analysis in detail, Matsuo et al. [9] reported that Japanese patients with normal SPECT images had an extremely good prognosis if their end-systolic volume and left ventricular ejection fraction (LVEF) also were normal. Additionally, Hashimoto et al. reported [10] prediction and risk stratification of cardiac events based on a combination of perfusion indices and cardiac function indices derived from gated myocardial perfusion SPECT, on the basis of J-ACCESS results indicating predictors of future cardiac events identified as age, DM, SSS categories, endsystolic volume, and LVEF. Hatta et al. [11] stratified the risk of cardiac events in patients with concurrent chronic kidney disease (CKD) with a combination of estimated glomerular filtration rate (eGFR), DM, and SSS on the basis of the J-ACCESS database. However, since the subjects enrolled in the J-ACCESS were patients suspected or confirmed CAD, the database did not sufficiently include high risk population having concurrent DM or CKD. Therefore, J-ACCESS II [12] and J-ACCESS III [13] were conducted in DM and CKD patients respectively, who had no history of CAD, to supplement insufficient data on the high risk population.
A series of the J-ACCESS provided risk stratification of cardiac events with nuclear cardiology, which is evidence for prognostic prediction in CAD patients in Japan. Such considerable evidence in the United States and Japan established the value of ischemia evaluation based on SPECT images in diagnosis and risk management of patients with CAD.
3. Cardiac event risk stratification using myocardial perfusion SPECT in CKD patients
The relationship between CKD patients and cardiovascular events has been noticed for a long time. It is well known that the incidence of all death and cardiovascular events increase with a decrease in eGFR and that many CKD patients die of cardiovascular events before starting hemodialysis therapy [14,15]. Therefore, screening of CAD is important and necessary for CKD patients [16,17]; myocardial perfusion SPECT is considered to be a suitable modality for screening CAD in CKD patients because those have frequent development of asymptomatic arteriosclerosis [18,19] and a risk of contrastinduced nephropathy [20,21]. In Japan, the multicenter J-ACCESS III [13] mentioned above was conducted in CKD patients without a history of CAD; the results demonstrated that three variables, i.e. SSS, eGFR, and LVEF, are significant predictors of cardiac events in such population. However, the J-ACCESS III database includes no data on severe CKD patients such as those requiring hemodialysis. Recently, Moroi et al. [22] conducted a multicenter nuclear cardiology study using 123I-BMIPP SPECT (B-SAFE: BMIPP SPECT Analysis for Decreasing Cardiac Events in Hemodialysis Patients) and have reported risk stratification of cardiac events based on BMIPP defect scores in hemodialysis patients. We also conducted a single-center, large-scale prognostic study using myocardial perfusion SPECT in a series of CKD patients including those with hemodialysis and reported a prognostic value of risk stratification of cardiac events based on SSS in each stage of CKD [23]. Furthermore, taking notice of a relationship between cardiac death and eGFR reduction within one year (ΔeGFR) in CKD patients, we stratified the risk of cardiac death using cut-off SSS (= 9) and ΔeGFR (= 10) based on results of receiver operating characteristic analysis and identified three variables, i.e. baseline eGFR, ΔeGFR, and SSS, as significant predictors of cardiac death [24]. In addition, Kaplan-Meier survival curves for non- CKD and CKD patients stratified with both cut-off SSS and ΔeGFR indicated significant risk stratification of cardiac death and CKD patients with SSS ≥ 9 and ΔeGFR ≥ 10 had the poorest prognosis in particular (Figure 1). This was a single-center study, but was the first large-scale prognostic study with myocardial perfusion SPECT in which the endpoint was cardiac death in Japanese CKD patients.
4. Usefulness of automated assessment with myocardial perfusion SPECT and cardiac event risk stratification
Usual ischemic quantification by nuclear cardiology is based on summed scores obtained from visual assessment of SPECT images. The visual assessment is intricate and requires a special skill or knowledge of image interpretation. A total perfusion deficit (TPD) proposed by Slomka et al. [25] is a new objective parameter, which is automatically calculated by the QPS (quantitative perfusion SPECT) software and represents both severity and extent of a defect in comparison with a normal database (NDB) created on the basis of American myocardial perfusion imaging data. In their study results, the TPD achieved performance better than or equivalent to visual quantification based on per-segment visual optimization of abnormality thresholds. In addition, automated quantitative assessment with the TPD was confirmed to provide highly correlated results with visual interpretation by three experts and to be more reproducible than the visual quantitative assessment for defect extent [26]. The automated quantitative assessment with the TPD has been treated as a reliable technique detecting CAD in the multicenter trial [27] and the American Society of Nuclear Cardiology imaging guideline 2010 for SPECT [28].
On the other hand, the automated assessment was barely recognized in Japan unlike the United States. One of the reasons for that is there was no validated Japanese NDB. In recent year the Japanese Society of Nuclear Medicine developed a Japanese NDB for myocardial perfusion images obtained from subjects with a low-likelihood of cardiac disease [29]. It was also unknown whether the TPD derived from the Japanese NDB has clinical significance. Therefore, using the TPD derived from the Japanese NDB, we automatically quantified SPECT images in Japanese CAD patients to compare with the conventional visual semi-quantitative assessment using the segmental defect scores, and reported that the automated quantification with the TPD was consistent with the visual quantification [30] and was reproducible [31]. Furthermore, we investigated the ability of the TPD derived from the Japanese NDB to predict future cardiac events in Japanese CAD patients and demonstrated that the predictive power of the automated quantification with the TPD is similar to that of the visual semi-quantification on the basis of the results of receiver operating characteristic analysis (Figure 2) and global chi-square comparison and that the risk of cardiac events can be stratified according to the TPD values (Figure 3) [32]. Considering the visual semi-quantification is subjective assessment and prognostic prediction with that is possibly different between expert and inexpert interpreters [33], the automated quantification with the TPD, in which such expert interpreters are unnecessary, allows stable prognostic prediction and is very useful for prognostic risk stratification of cardiac events.
5. Decision of therapeutic strategy for CAD on the basis of ischemic volumes obtained from myocardial perfusion SPECT and prediction of cardiac events after treatment
Estimation of the amount of myocardial ischemia is extremely important for decision of a therapeutic strategy in patients with stable angina pectoris. Hachamovitch et al. [34] compared a prognosis after revascularization with that after medical therapy alone and evaluated a relationship between the prognosis and the amount of ischemia (% of total myocardium ischemic) estimated with myocardial perfusion SPECT. Cardiac death was less after early revascularization than medical therapy alone in patients with >10% ischemic myocardium estimated with myocardial perfusion SPECT. On the other hand, patients with ≤10% ischemic myocardium experienced a good prognosis after optimal medical therapy alone in comparison with revascularization. Moroi et al. [35] matched propensity scores extracted from J-ACCESS database to examine a relationship between cardiac event rates and the amount of ischemic myocardium, and reported that early revascularization possibly leaded to a good prognosis in Japanese patients with >10% ischemic myocardium estimated with myocardial perfusion SPECT. According to such reports, we consider that a good prognosis may result from a therapeutic strategy determined on the basis of the severity of ischemic myocardium estimated with myocardial perfusion SPECT before treatment. A relationship actual ischemic reduction due to treatment and the occurrence of cardiac events after the treatment was evaluated in the COURAGE Trial Nuclear Substudy [27]. In patients with ≥10% ischemic myocardium estimated with myocardial perfusion SPECT before treatment, ≥5% ischemic reduction after the treatment resulted in a good prognosis in comparison with <5% ischemic reduction. Additionally, ischemic reduction observed on SPECT images was greater with PCI plus optimal medical therapy than with optimal medical therapy alone. In Japan, the J-ACCESS IV trial [36], which is similar to the design of the COURAGE Trial Nuclear Substudy, has been started in order to collect data prospectively. Prior to that, we conducted a retrospective pilot study to investigate a relationship between the ischemic reduction and the onset of cardiac events after treatment in Japanese patients with CAD. A good prognosis was observed in patients with than without ≥5% ischemic reduction and in patients without than with residual ischemia [37]. Our results were similar to those in the COURAGE Nuclear Substudy [27] and the appropriate cut-off value for ischemic reduction was also “5%”. Therefore, it may be a consistent reliable target value for reduction of ischemia where revascularization is performed to improve prognosis among any racial patient with CAD.
6. Management of CAD on the basis of future cardiac event rates estimated by risk scores
The major strong point of nuclear cardiology is distinctly superior quantification of ischemic myocardium due to coronary arterial lesions. Combination of ischemic indices obtained from SPECT images and other risk factors associated with major cardiac events (MCEs) allows us to predict the risk of future MCEs. Nakajima et al. [38] published a Heart Risk Table in 2012, which is an equation for estimation of MCEs within three years. The equation consists of five independent predictors and weight coefficients for those, which are age, presence/absence of DM, LVEF, SSS categories, and eGFR extracted by the multivariate logistic regression analysis on the basis of the J-ACCESS database (n = 2,543). Only inputting values for those five risk factors mentioned above to the Heart Risk Table, we can predict the risk of MCEs within three years in the Japanese patient. Estimation of cardiac event risk prior to treatment leads to patient’s motivation for accepting positively the treatment. However, data of MCEs used to prepare the Heart Risk Table were obtained from 27 patients with cardiac death, 25 with non-fatal myocardial infarction, and 64 with heart failure requiring hospitalization; 55% of the MCEs was occupied by heart failure. Consequently the Heart Risk Table is a regression equation suitable to predict the risk of severe heart failure in particular of MCEs.
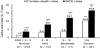
Therefore we performed a three-year prognostic follow-up study in 2,579 patients who underwent myocardial perfusion SPECT in our hospital to prepare a regression equation suitable to predict the risk of MCEs excepting heart failure and reported our original cardiac event risk score (CERS) [39]. The following is a description of the creation of the CERS according to our report. During the three-year follow-up, 171 (6.6%) of patients experienced MCEs including cardiac death (n = 78), non-fatal myocardial infarction (n = 30), and unstable angina pectoris (n = 63). Figure 4 shows the relationship between cardiac death or MCE rates during the three-year follow-up and baseline SSS categories. The MCE rate significantly increased in proportion to SSS. The cardiac death rate was not different among SSS normal, mildly, and moderately abnormal groups but significantly higher in the severely abnormal group (SSS ≥ 13); that was consistent with the results obtained from the past study. Table 1 shows independent predictors for MCEs and weight coefficients for those, which were extracted by the multivariate logistic regression analysis. According to our results, predictors for MCEs were four factors consisting of age, presence/absence of DM, SSS, and eGFR, and predictors for only cardiac death were four factors consisting of age, stress LVEF, SSS, and eGFR; the predictors were different between MCEs and only cardiac death. An increase in the number of the predictors combined made the global chi-square value rise significantly (Figure 5), which resulted in improvement of predictive precision for MCEs or only cardiac death. On the basis of these results, we prepared the following two original CERSs providing the risk (%) of MCEs and cardiac death within three years:
MCE risk (%/3 years) = 1/{1 + Exp[–(–3.176 + 0.018 × age + 0.602 × DM – 0.022 × eGFR + 0.051 × SSS)]} × 100
Cardiac death risk (%/3 years) = 1/{1 + Exp[–(–2.602 + 0.031 × age – 0.031 × eGFR + 0.038 × SSS – 0.029 × stress LVEF)]} × 100
where age is given as years old, DM is “1” for presence or “0” for absence, eGFR as mL/min/1.73 m2, SSS as continuous variables, and LVEF as %.
Table 2 shows the risks of MCEs and cardiac death within three years estimated by the original CERSs in some patients. The MCE predicting model indicates estimated MCE risks within three years based on the data on age, DM, eGFR, and SSS and the cardiac death predicting model indicates estimated cardiac death risk within three years based on the data on age, eGFR, SSS, and stress LVEF. The risk of MCEs resulting from acute coronary syndrome is largely affected by the presence of DM. On the other hand, the risk of cardiac death is largely affected by cardiac dysfunction-related index (low LVEF). Those are understandable results. This was the first report demonstrating that the predictors for the risk of cardiac events are rather different between MCEs and cardiac death in Japanese patients.
7. Conclusion
Nuclear cardiology is a useful modality for prediction and risk stratification of cardiac events. Quantitative ischemic indices derived visually or automatically from myocardial perfusion SPECT data provide risk stratification of cardiac events, which is helpful for improvement of prognosis in high risk population such as patients with DM or CKD. The risk score created with data obtained from gated myocardial perfusion SPECT should be effectively used in daily clinical practices because it is important to explain benefit and risks to CAD patients before decision of a therapeutic strategy.
Competing Interests
The authors declare that they have no competing interests.